BioPathogenix
Powering the
Diagnostic Engine
BioPathogenix is Omni Genomix's exclusive reagent and laboratory supply partner — providing clinical-grade molecular reagents, diagnostic instruments, pathogen panels, and consumables that power the entire testing infrastructure.

Laboratory Supply. Reinvented.
BioPathogenix provides everything your molecular diagnostics laboratory needs — from high-purity reagents to automated workflow solutions — all integrated with the Omni Genomix ecosystem.
Molecular Reagents
High-purity PCR reagents, extraction kits, and amplification enzymes optimized for clinical molecular diagnostics and pathogen detection.
Diagnostic Instruments
Next-generation molecular diagnostic instruments including real-time PCR systems, sample prep automation, and rapid detection platforms.
Pathogen Panels
Comprehensive multiplex pathogen detection panels covering respiratory, GI, STI, and wound infection targets with high sensitivity and specificity.
Quality Controls
Third-party verified quality control materials and proficiency testing panels to ensure laboratory accuracy and regulatory compliance.
Lab Consumables
Full catalog of laboratory consumables — pipette tips, plates, tubes, swabs, and transport media for specimen collection and processing.
Automation Solutions
Workflow automation systems that integrate with PrymDx LIMS for hands-free sample processing, reducing human error and increasing throughput.
From Assessment to Continuous Supply.
BioPathogenix doesn't just ship reagents — they partner with your laboratory to optimize workflows, reduce waste, and ensure uninterrupted testing operations.
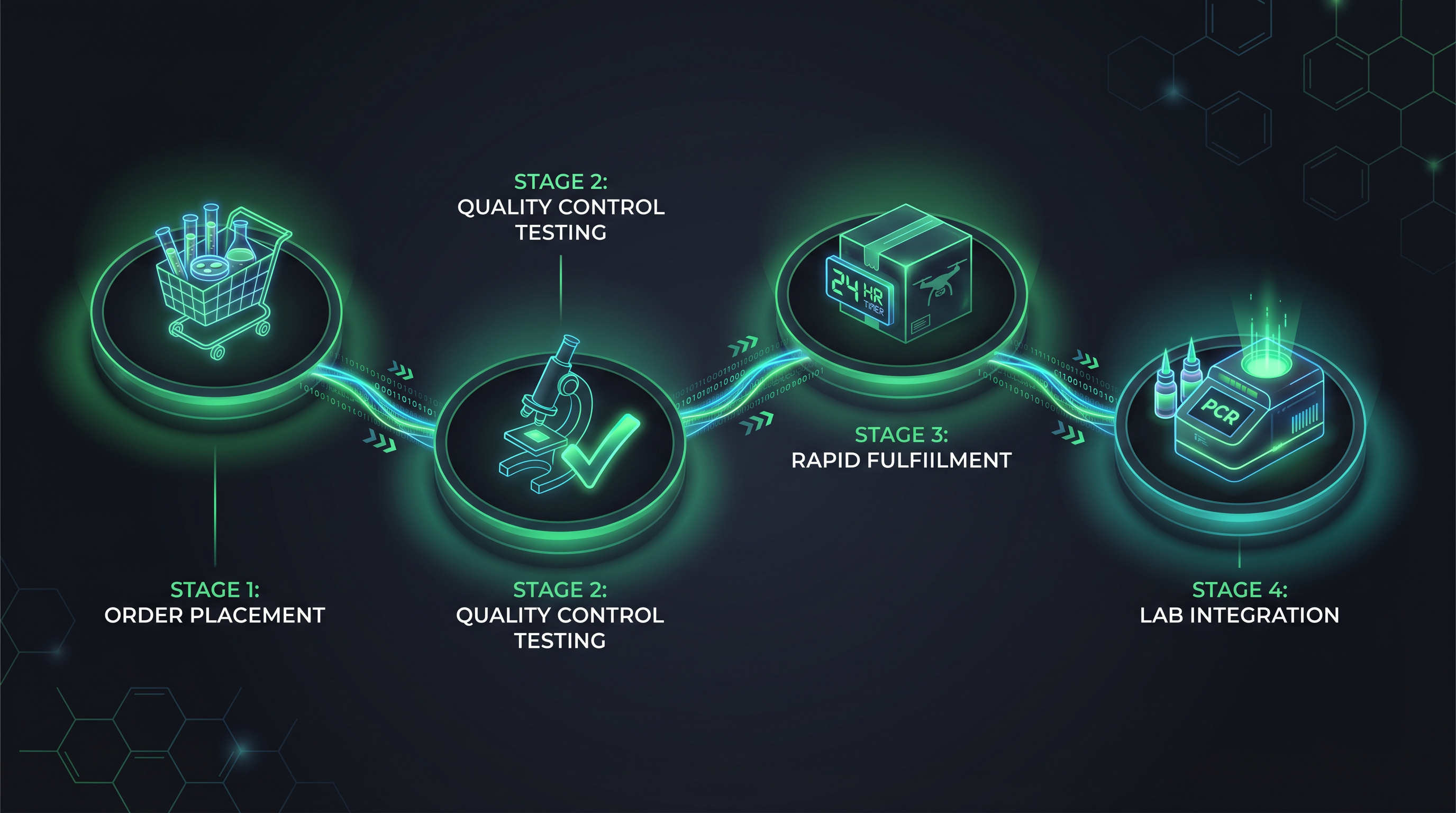
Assess Needs
BioPathogenix consultants assess your laboratory's testing menu, volume, and workflow requirements to recommend optimal reagent configurations.
Configure & Ship
Custom reagent kits and supplies configured for your specific instruments and protocols. Cold-chain shipping ensures stability.
Integrate
Reagents integrate seamlessly with PrymDx LIMS for automated lot tracking, expiration alerts, and quality control documentation.
Support & Reorder
Ongoing technical support, auto-replenishment programs, and dedicated account management keep your lab running without interruption.
Not Just a Supplier. A Lab Partner.
Traditional reagent distributors ship boxes. BioPathogenix integrates with your laboratory infrastructure — automating inventory, ensuring quality, and optimizing every dollar spent on consumables.
Clinical-Grade Reagents
Every reagent is manufactured under ISO 13485 quality management systems. Lot-to-lot consistency ensures reproducible results across thousands of patient samples.
Omni Genomix Integrated
BioPathogenix is the exclusive reagent and supply partner for the Omni Genomix ecosystem. Direct integration with PrymDx LIMS enables automated inventory and QC tracking.
Cold-Chain Logistics
Temperature-sensitive reagents ship via validated cold-chain logistics with real-time monitoring. Guaranteed stability from warehouse to your laboratory bench.
Cost Optimization
Bulk pricing, auto-replenishment programs, and waste reduction through precise volume forecasting. Labs save 15-25% compared to traditional distribution channels.
Nationwide Distribution
Strategically located distribution centers enable next-day delivery across the continental US. Emergency same-day shipping available for critical reagent needs.
Technical Support Team
Dedicated field application scientists provide on-site training, troubleshooting, and protocol optimization. Your lab's success is their mission.
Trusted by Leading Labs.
"BioPathogenix reagents have been rock-solid for our molecular diagnostics program. The integration with PrymDx LIMS for lot tracking alone saves us hours of manual documentation."
"The auto-replenishment program eliminated our stockout issues completely. We haven't had a single testing delay due to reagent availability since switching to BioPathogenix."
"The quality consistency is what sets them apart. When you're running thousands of patient samples, you need reagents you can trust. BioPathogenix delivers that confidence."
Key Product Categories
From molecular reagents to automation solutions — everything your laboratory needs to deliver accurate, timely diagnostic results.
Molecular Reagent Kits
Complete PCR reagent systems including master mixes, primers, probes, and extraction buffers optimized for clinical molecular diagnostics.
Multiplex Pathogen Panels
FDA-cleared and LDT-validated multiplex panels for respiratory, gastrointestinal, STI, and wound infection pathogen detection.
Diagnostic Instruments
Real-time PCR thermocyclers, automated extraction systems, and rapid molecular platforms designed for high-throughput clinical laboratories.
QC & Proficiency Materials
Third-party verified quality control materials, proficiency testing panels, and calibration standards for regulatory compliance and accreditation.
Collection & Transport
Specimen collection devices, transport media, swabs, and pre-analytical supplies designed for optimal sample integrity from collection to analysis.
Automation & Integration
Laboratory automation solutions that connect directly with PrymDx LIMS for seamless sample tracking, result reporting, and inventory management.
The Supply Layer of the Omni Genomix Stack
BioPathogenix powers the reagent and consumable infrastructure that every other layer of the Omni Genomix ecosystem depends on — from specimen collection to molecular analysis.
Omni Genomix Capabilities
Ecosystem Partners
Real Results from Real Practitioners
See how practitioners across the country are transforming their practices with BioPathogenix — measured in revenue, efficiency, and patient outcomes.
Dr. Thomas Reed
PhD, MT(ASCP) — Lab Operations Director
The Challenge
Managing 14 different reagent suppliers with separate ordering systems, inconsistent quality, and unpredictable lead times. Three stockout events in 6 months caused $120K in lost revenue and delayed 2,100 patient results.
The Implementation
Consolidated all reagent procurement through BioPathogenix single-source platform. Automated predictive reordering based on consumption patterns, with guaranteed 48hr emergency fulfillment and lot-specific QC certificates.
Measured Outcomes
Stockout Events
Zero downtime
Supplier Count
-93% complexity
QC Failure Rate
-86% failures
Procurement Hours
-88% admin time
"We had three stockouts in 6 months that cost us $120K and delayed thousands of patient results. Since switching to BioPathogenix, we've had zero stockouts in 18 months. The predictive reordering is almost eerie — supplies arrive before we even realize we're running low."
— Dr. Thomas Reed, PhD, MT(ASCP) — Lab Operations Director
Dr. Karen Mitchell
MD, FCAP — Pathology Director
The Challenge
Lot-to-lot reagent variability causing 15-20 QC failures per month, each requiring repeat testing and delayed results. Cold-chain shipping failures wasted $8K/month in expired reagents. CAP inspection preparation took 4 weeks annually.
The Implementation
Migrated to BioPathogenix with ISO 13485-certified reagents, temperature-monitored shipping with real-time GPS, automated lot transition protocols, and digital compliance vault for instant inspection readiness.
Measured Outcomes
QC Failures/Month
-89% failures
Reagent Waste Cost
-97% waste
CAP Prep Time
99.9% faster
Repeat Testing Rate
-87% repeats
"Our QC failures dropped from 18/month to 2/month — and those two were operator error, not reagent quality. The cold-chain monitoring alone saved us $96K/year in wasted reagents. When CAP showed up for a surprise inspection, we pulled every document they needed in under 60 seconds."
— Dr. Karen Mitchell, MD, FCAP — Pathology Director
Lab Supply Chain, Solved
Reagent shortages, quality inconsistencies, and supply chain disruptions cost labs millions in downtime and failed assays. BioPathogenix eliminates supply uncertainty.
Reagent stockouts halt lab operations and delay patient results for days
Predictive inventory management with automated reorder triggers, safety stock algorithms, and guaranteed 48hr emergency fulfillment
Zero stockout-related lab downtime in the past 18 months
Lot-to-lot reagent variability causes QC failures and repeat testing
ISO 13485-certified manufacturing with lot-specific QC certificates, pre-validated performance data, and automated lot transition protocols
99.4% first-pass QC acceptance rate across all reagent lots
Labs juggle 10-15 different reagent suppliers with separate ordering systems
Single-source reagent platform covering molecular, immunoassay, and clinical chemistry — one vendor, one invoice, one quality standard
80% reduction in procurement complexity and vendor management
Cold-chain reagent shipping failures waste expensive materials and delay testing
Temperature-monitored shipping with real-time GPS tracking, validated packaging for 72hr cold-chain integrity, and automatic replacement for any excursion
99.8% cold-chain compliance with zero-cost replacement guarantee
New assay development requires months of reagent sourcing and validation
Custom reagent formulation service with dedicated R&D team, rapid prototyping, and accelerated validation protocols for novel assays
New reagent formulations validated and production-ready in 4-6 weeks
Regulatory documentation for reagents is scattered across multiple systems
Digital compliance vault with automated SDS management, expiration tracking, and one-click regulatory submission packages for CLIA/CAP inspections
Inspection-ready documentation available in under 60 seconds
How Data Flows Through BioPathogenix
Every partner in the Omni Genomix ecosystem is connected. Data flows seamlessly between layers — no silos, no manual handoffs.

BioPathogenix
CurrentData Transferred
Reagent kits
QC certificates
Lot tracking data
Expiry manifests
ISO 13485 compliance
Data Transferred
Inventory levels
Reorder triggers
Batch traceability
Calibration standards

Ready to Optimize Your Lab Supply Chain?
Join the Omni Genomix ecosystem and get access to clinical-grade reagents, automated inventory management, and dedicated technical support.

