Spot Dx
Home Testing for
Health Brands
Spot Dx provides everything you need to offer your users lab-analyzed blood, urine, and saliva tests with at-home sample collection. White-label kits, developer API, and HIPAA-compliant results — launch in days, not months.

At-Home Testing. Your Brand.
Spot Dx is a turnkey solution for launching at-home lab testing under your own brand. White-label kits, developer-first API, and CLIA-certified lab processing — everything you need to offer diagnostics to your users.
At-Home Collection Kits
White-label at-home collection kits for blood, urine, and saliva testing. Patients collect samples at home and ship to partner labs using prepaid mailers.
White-Label Branding
Launch with packaging and digital tools that match your brand. Patients see your logo, your colors, and your experience — not a third-party lab company.
Developer-First API
Comprehensive API and developer documentation for seamless integration. Order kits, track shipments, and receive results programmatically — launch in days, not months.
HIPAA Compliant
End-to-end HIPAA compliance from kit ordering to results delivery. Enterprise-grade security protects patient data at every step of the diagnostic journey.
Multi-Specimen Support
Blood, urine, and saliva collection kits available. Support for a wide range of diagnostic panels — from metabolic and hormone to specialty and advanced biomarkers.
Medically-Vetted Interpretations
Results include normal ranges and medically-vetted interpretations. Integrate results directly into your customer-facing app with rich, actionable data.
Kit. Collect. Results. Turnkey Simple.
From ordering kits to delivering results, Spot Dx handles the entire at-home testing operation. Focus on what you do best — let Spot handle the logistics, lab processing, and compliance.
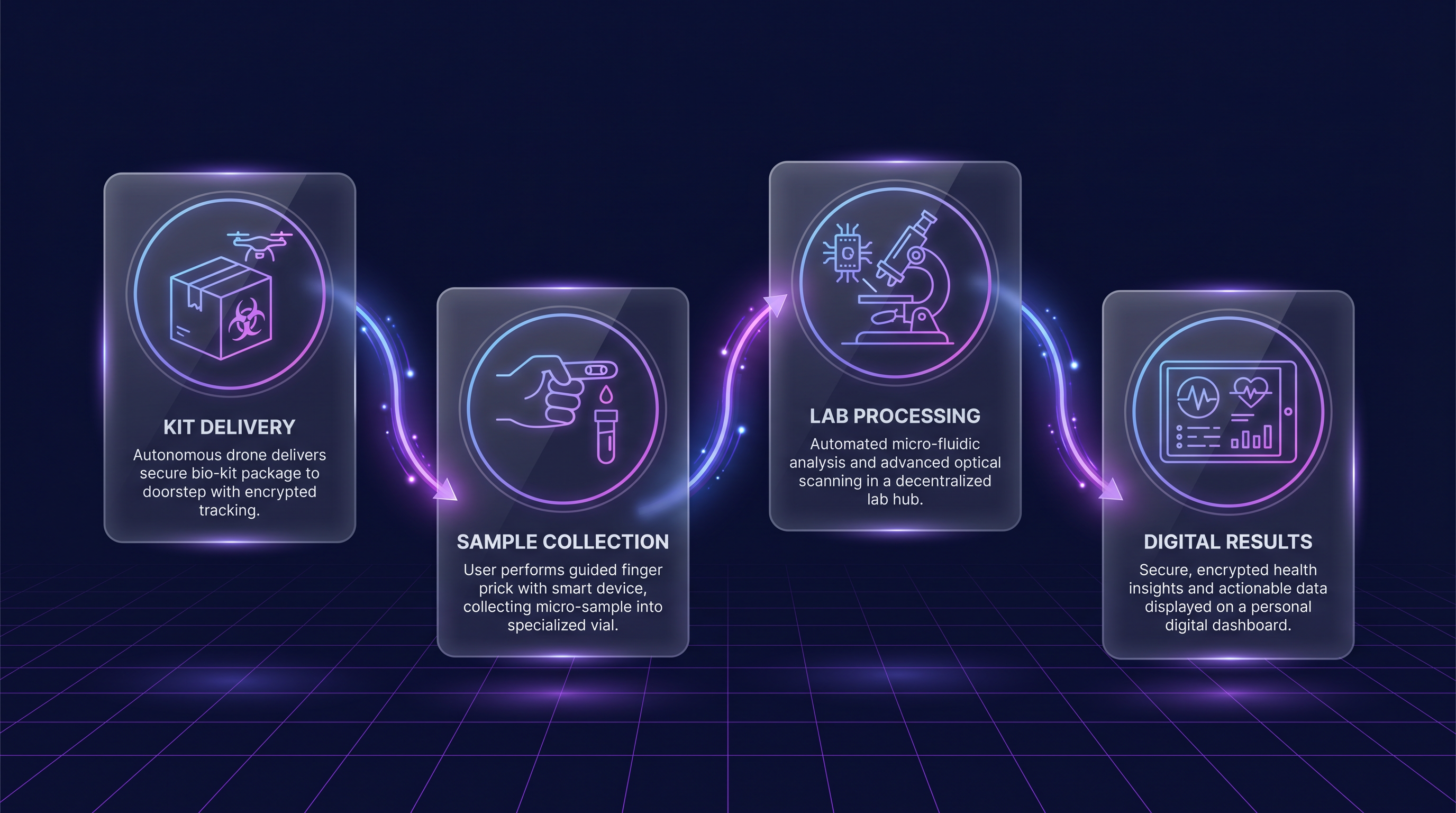
Order Kits
Send an at-home collection kit to your user. Some states require a doctor's order — Spot handles this automatically.
Collect Sample
Patient collects a blood, urine, or saliva sample at home following simple instructions included in the kit.
Ship to Lab
Patient sends the sample to a partner lab using the provided prepaid mailer. Full chain-of-custody tracking.
Get Results
Results with normal ranges and medically-vetted interpretations are delivered through the API or patient portal.
Not Just Testing. A Brand Extension.
Traditional lab testing requires patients to visit clinics and interact with unfamiliar brands. Spot Dx lets you own the entire experience — from the kit in their mailbox to the results in their app.
True White-Label Experience
Every touchpoint — from the collection kit packaging to the digital results portal — carries your brand. Patients never see Spot Dx; they see the brand they trust.
Omni Genomix Integrated
Spot Dx is natively integrated with the Omni Genomix ecosystem. Practitioners can order white-label collection kits directly through the platform.
Developer-First Architecture
Comprehensive REST API with webhooks, status tracking, and result delivery. Developer documentation makes integration straightforward — launch in days, not months.
Demonstrate Product Efficacy
Run efficacy studies at a fraction of the cost. Use lab-analyzed results to promote your products with evidence-based claims that build consumer trust.
Nationwide Lab Network
Partner labs across the United States process specimens with CLIA certification and CAP accreditation. Consistent quality regardless of patient location.
Personalize Health Products
Tailor supplements, fitness programs, and meal plans to each user's specific biomarker results. Track progress over time with repeat testing.
Brands Trust Spot Dx.
"Spot Dx let us launch at-home testing under our own brand in less than two weeks. The API integration was seamless and our users love the experience."
"We use Spot Dx to personalize supplement recommendations based on real lab data. It's transformed our customer retention and average order value."
"Running efficacy studies used to cost us a fortune. With Spot Dx, we can demonstrate product efficacy with real biomarker data at a fraction of the cost."
Key Product Integrations
Every component of the Spot Dx platform is purpose-built for white-label at-home testing and natively connected to the Omni Genomix ecosystem.
At-Home Collection Kits
Core Product
White-label blood, urine, and saliva collection kits shipped directly to patients. Custom packaging with your brand, simple instructions, and prepaid return mailers.
Developer API
Integration Platform
RESTful API for ordering kits, tracking shipments, and receiving results programmatically. Webhook notifications for real-time status updates and result delivery.
White-Label Portal
Brand Experience
Customer-facing digital portal branded with your logo, colors, and design language. Patients view results, track orders, and manage their testing history under your brand.
Lab Processing Network
Specimen Analysis
CLIA-certified and CAP-accredited partner laboratories process all specimens. Results include normal ranges and medically-vetted interpretations for clinical accuracy.
Efficacy Studies
Evidence Platform
Run product efficacy studies using real biomarker data at a fraction of traditional costs. Demonstrate evidence-based claims that build consumer trust and regulatory confidence.
Health Personalization
Product Tailoring
Use lab results to personalize supplements, fitness programs, meal plans, and health products for each user. Track progress with repeat testing over time.
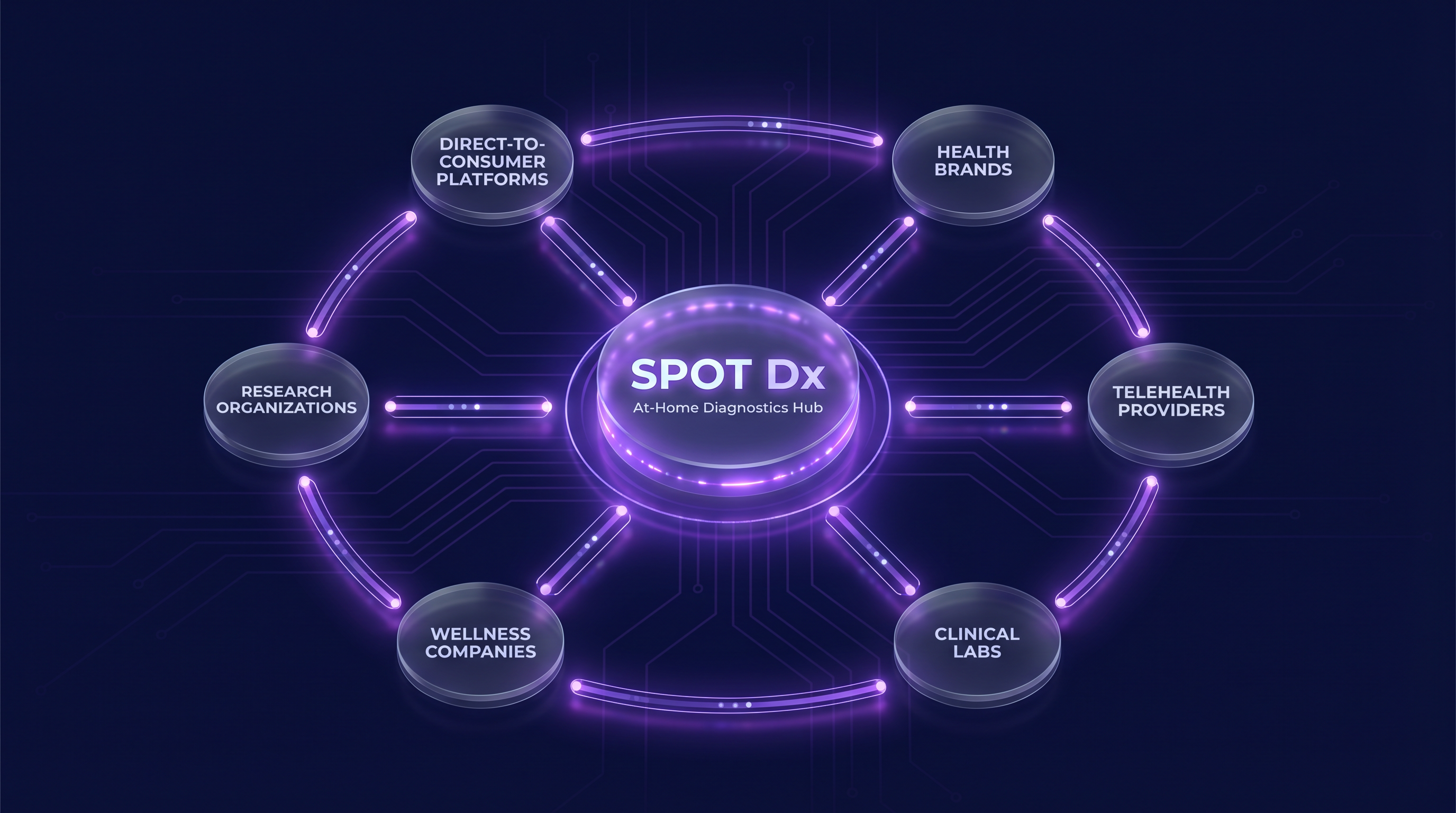
Natively Integrated with Every Omni Genomix Service
Spot Dx doesn't exist in isolation. It's connected to every service in the Omni Genomix ecosystem — from nutrigenomics testing and telehealth consultations to pharmacy fulfillment and LIMS processing.
Omni Genomix Capabilities
Partner Brands & Networks




Real Results from Real Practitioners
See how practitioners across the country are transforming their practices with Spot Dx — measured in revenue, efficiency, and patient outcomes.
Dr. Amanda Foster
DO, IFMCP — Functional Medicine
The Challenge
100% virtual practice with patients in 12 states. Couldn't scale because each state had different lab logistics. Patients were confused by collection instructions, and 45% of at-home kits were returned with insufficient samples.
The Implementation
Launched branded 'Root Cause Collection Kits' through Spot Dx with custom panel configurations, state-compliant consent workflows, and patented collection devices with video-guided instructions.
Measured Outcomes
Geographic Reach
+300% coverage
Kit Success Rate
+75% completion
Monthly Revenue
+127% growth
Patient Capacity
+133% scale
"I went from serving patients in 3 states to 12 states in 60 days — Spot Dx handled all the regulatory complexity. My branded kits have a 96% success rate because the collection devices are literally foolproof. Patients love the experience and it reinforces my brand at every touchpoint."
— Dr. Amanda Foster, DO, IFMCP — Functional Medicine
Dr. David Kim
MD — Preventive & Longevity
The Challenge
Wanted to launch a direct-to-consumer preventive testing brand but couldn't justify the $2M+ investment in lab infrastructure. Needed white-label kits, fulfillment, and regulatory compliance across all 50 states.
The Implementation
Built entire DTC testing brand on Spot Dx infrastructure — custom-branded kits, automated fulfillment from 3 distribution centers, 50-state compliance, and API integration with his patient portal.
Measured Outcomes
Launch Investment
-98% capital needed
Monthly Orders
From zero to scale
Gross Margin
Premium economics
Time to Market
-92% faster
"I launched a DTC testing company with zero lab infrastructure investment. Spot Dx handles everything from kit manufacturing to fulfillment to compliance. We went from concept to 1,200 monthly orders in 8 months. The unit economics are better than if I'd built my own lab."
— Dr. David Kim, MD — Preventive & Longevity
Scaling Diagnostics Without Infrastructure
Building a branded diagnostic program traditionally requires millions in lab infrastructure, regulatory expertise, and logistics. Spot Dx eliminates all of it.
Practitioners want branded lab testing but can't afford to build their own lab
White-label collection kit platform with custom branding, practitioner-specific panels, and turnkey fulfillment — no lab infrastructure needed
Launch a branded testing program in under 30 days
At-home collection kits have high failure rates due to poor patient instructions
Patented collection devices with foolproof design, QR-linked video instructions, and built-in specimen quality indicators
96% successful self-collection rate on first attempt
Kit logistics (inventory, shipping, returns) consume operational bandwidth
Full kit lifecycle management — warehousing, on-demand fulfillment, prepaid return shipping, and automated reorder triggers
Zero operational overhead for kit logistics
Regulatory requirements for at-home collection vary by state and test type
Built-in regulatory compliance engine handling state-by-state requirements, informed consent workflows, and physician authorization routing
Compliant operation across all 50 states without legal counsel
Practitioners can't track which patients have completed their at-home kits
Real-time kit status dashboard showing shipped, activated, returned, and processing stages with automated patient reminders
85% kit return rate (vs. 55% industry average)
Custom panel development requires months of R&D and validation
Pre-validated panel library with 200+ biomarker combinations, plus rapid custom panel development with 2-week validation turnaround
New panels live in 14 days vs. 6+ months traditionally
How Data Flows Through Spot Dx
Every partner in the Omni Genomix ecosystem is connected. Data flows seamlessly between layers — no silos, no manual handoffs.

Spot Dx
CurrentData Transferred
White-label kit ID
Specimen barcode
Collection instructions
Return logistics
Data Transferred
Kit activation data
Patient registration
Sample integrity flags
Shipping manifests
Ready to Launch White-Label Testing?
Join the Omni Genomix ecosystem and offer your patients branded at-home testing through Spot Dx. Developer-first API, HIPAA-compliant processing, and a turnkey solution that launches in days.